Introduction
Managing wound infection is an essential component for success in chronic wound healing. Preventing the wound from infection and keeping the wound free from bacteria is a difficult task to archive. In chronic wound infection or deep wound infection, the usual treatment is surgical debridement accompanied by systemic antibiotics for delivering antibiotics to the infection site [1-4].
However, many chronic wounds have inadequate blood supply around the wounds due to underlying disease, tissue necrosis, radiation, and etc. For examples most diabetic patients commonly have poor blood supply on the lower extremity. This raises a question on systemic antibiotic those most likely are less efficient because they cannot penetrate into the wound area due to compromise of blood supply. Therefore, topical antiseptic or antiseptic wound dressing may be sometime more useful because the systemic antibiotics are not very practical. Furthermore, over usage or long-term treatment with topical antiseptics can cause cytotoxicity to host cells especially on fibroblasts, keratinocytes, and endothelial cells which are significantly taking part in wound healing process, causing wound healing process to slow down. In addition, long-term dressing with antiseptics frequently causes irritation or allergic dermatitis. Therefore, topical antibiotics that can sustain their effect in an extended period are often needed to replace topical antiseptics, so that wound healing process does not disrupt.
Fibrin sealant is a well-known hemostatic agent that has been known over the past decade. Fibrin sealants consist of fibrinogen and thrombin components, both of which are isolated from human plasma [3]. It has remarkable properties, that turn it into a net-like structure of a fibrin sealant clot with both fibrinogen and thrombin [5], improving wound healing. It is biodegradable and also promotes wound healing without the need to remove it. Since the fibrin sealant is a gel-like liquid, it can be applied to any small space or gap, making it a close to ideal material as a local antibiotic drug delivery vehicle.
Although previously published reports have presented clinical trials and effect of a sustained release of the antimicrobial agents [6,7], the optimal drug concentration and duration for sustained effect against bacteria are still unknown. The present study evaluates the effect of antibiotic-saturated fibrin sealant against Methicillin-Resistant Staphylococcus Aureus (MRSA, ATCC33591; Microbiology Laboratory, Authors’ Institution, Seoul, Korea) by adjusting the concentration of the antibiotic as well as the duration exposure.
Methods
Medium and reagents
Fibrin sealants (Tisseel®; Baxter AG, Wien, Austria), Dulbecco’s Modified Eagle and Medium/Ham’s F-12 nutrient (DMEM/F-12; Gibco; Grand Island, NY, USA), Mueller-Hinton agar (MHA-100, Korea Medical Holdings, Seoul, Korea) and blood agar (Infusion Agar, Asan Pharmceutical, Seoul, Korea) were purchased separately from their pharmaceutical companies. The antimicrobial drug utilized in this study was Teicoplanin (Ildong Pharm. Co., Seoul, Korea).
Microorganisms
The MRSA bacteria was placed on blood agar and grown for 24 hours in an incubator with 5% CO2 incubator at 37ºC. A single colony was harvested into a glass tube with 3 mL of distilled water and was measured using a Vitek colorimeter to be at a concentration of approximately 1×105 colony-forming units/mL. A thin layer of MRSA was then gently scrubbed and allowed to achieve full coverage of the inoculated on the MHA surface, which was then allowed to dry.
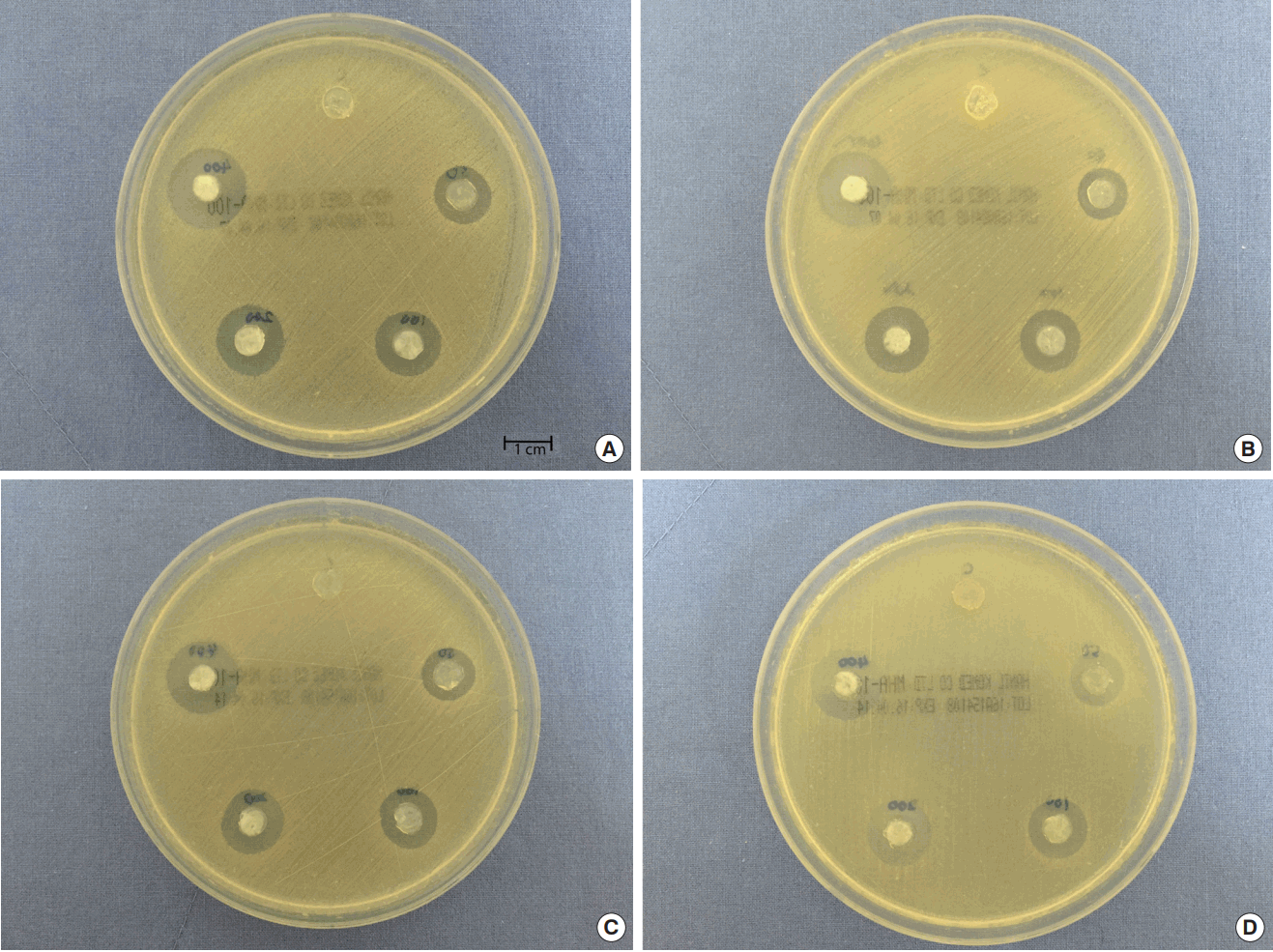
Antibiotic-saturated fibrin disc test
The teicoplanin antibiotic was impregnated in the fibrin disc at concentrations of 50 μg/mL, 100 μg/mL, 200 μg/mL, 400 μg/mL, and control (0 μg/mL) in 96 cell culture cluster wells to shape it into a disc-like shape (3 mm×5 mm). The fibrin discs were moved into a 24-cell culture well with DMEM/F-12 to obtains the transuded condition, and the samples were incubated at 37ºC in CO2 incubator for 7, 14, 21, and 28 days. After the incubation period, the antibiotic-saturated fibrin discs were transferred to MHA surface scrubs with MRSA (n=5).The antimicrobial susceptibility testing was carried out according to Clinical and Laboratory Standards Institution (CLSI) document M100-S23 (ISBN 1-56238-865-7 [Print]; ISBN 1-56238-866-5 [Electronic]) which is 14 mm in diameter for teicoplanin antibacterial effect against MRSA.
Filter paper test
The antibiotic disc was removed from the cell culture cluster well, leaving behind DMEM/F-12 with antibiotic that had been released from the disk at 7, 14, 21, and 28 days. Immediately after, filter paper was cut into round sample with a 5 mm diameter to match the antibiotic disc and was inserted into the cluster well. The filter papers were incubated for 24 hours at 37ºC in the CO2 incubator, and the filter papers were inoculated with MHA scrubbed with the MRSA strain. After 24 hours, the diameter of the non-bacterial inhibition zone on the filter paper was measured, and the mean was recorded (n=5).
Results
Antibiotic-saturated fibrin disc test
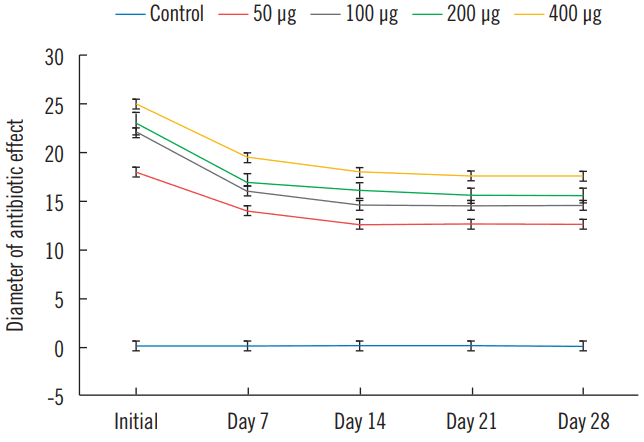
The antibiotic-saturated fibrin disc showed a greatest antibacterial activity during the first week. All groups except a control group demonstrated sustained effect until the fourth week (Figs. 1, 2, and Table 1).The antibacterial effect sharply decreased after the first week, but continued to exhibit a regular antibacterial effect up to the fourth week. However the 50 μg/mL groups fell below suitability within 14 days. Other groups maintained their effect for 28 days, and these 3 groups showed suitable effect.
Filter papers test
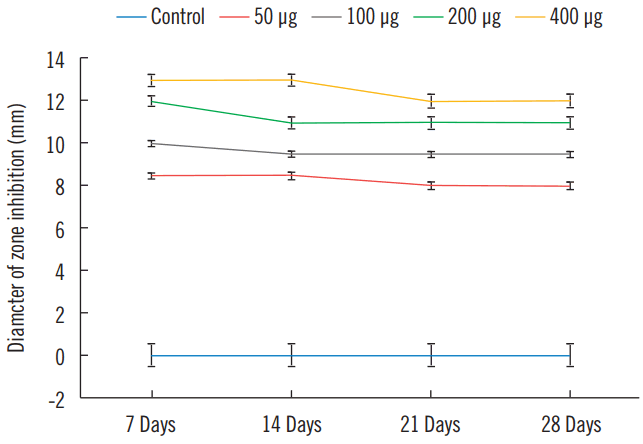
After 24 hours of incubation in DMEM/F-12 (after the antibiotic-saturated fibrin disc had been removed) at 37ºC in a CO2 incubator, all different concentrations showed stable, constant release with noticeable antibiotic activity. All four inhibition measurements induced by the filter paper absorbent against MRSA demonstrated a prolonged and sustained release (Figs. 3, 4, and Table 2).
Discussion
Chronic diabetic foot ulcer has been a major cause of lower limb amputation, and vascular system failure prevents systemic antibiotic form reaching an infection site, causing severe problems that lead to an amputation. Surgical debridement is one of the best options to remove pathologic tissues and may achieve an infection-free wound site. Additionally, it is usually necessary to maintain a high concentration of the appropriate antibiotic at the wound site for a sufficiently long time [2].
Poly (methyl methacrylate) antibiotic-impregnated beads are commonly used as prophylactic for local acute or chronic infection. However, poly (methyl methacrylate) only acts as a delivery vehicle for local drug delivery and has to be removed when the antibiotic effect is exhausted [8]. As an alternative, fibrin sealants can also be used as well as to support tissue regeneration or vehicles for delivery for tissue regeneration in chronic wounds [5,9,10]. There are many reports on the encapsulation and release of antibiotics from fibrin sealant [11,12], which show it potential as a biocompatible, biodegradable drug delivery system because sealant clots adhere to the target tissue and allow for controlled release of the drug over an extended period of time [13]. In this study, we used a fibrin sealant saturated with teicoplanin to assess its potential therapeutic activity against MRSA.
In previous studies, some hot topics debate about the moist wound and dry wound healing process. It turned out that moist wound healing has a better outcome than dry wound. In moist condition, wounds tend to heal much faster than in dry wound. Scabs were commonly forming in a dry wound, and it can lock out the oxygen provided from the environment to the wound side especially in inadequate blood supply chronic diabetic foot patients. However, fibrin sealants are a combination of fibrinogen and thrombin that extracted from blood serums. It because of it gel-like properties, fluid can be lock within after both fibrinogen and thrombin are mixed and from fibrin sealants. This is an addition ideal property that fibrin sealants provided to maintain moist to a chronic open wound.
The range of a safe dose for the topical antibiotic administration is important issue to be considered [8]. In this study, we have demonstrated treatment effect with teicoplanin at the given concentrations. MRSA is a resistant strain that is commonly manifested in a chronic infection. In the meantime, we have tried to control the level of the teicoplanin concentration and determine the limitations when controlling the concentration. The fibrin sealant has shown that it can maintain a constant drug release rate. We monitored the drug concentration, but the release rate from the fibrin sealant was unexpected. This in vitro study shows optimal, long-lasting effect for the drugs released for over 28 days. Although the development of drug resistance is a serious concern to surgeons, we do not believe that this method of topical application of a controlled-release formulation of would exacerbate this problem [13].
In CLSI document M100-S23 the antibiotic susceptibility test indicated that 14 mm are the susceptibility value of teicoplanin antibacterial effect for MRSA. From the result shown (Fig. 2), 100 μg/mL, 200μg/mL and 400 μg/mL have surpassed the antimicrobial susceptibility test. Therefore, the optimal concentration from this study can be 100 μg/mL. We did mention that developments of drug resistance are the concern for all healthcare parties; 100 μg/mL should be used as an optimal concentration for topical antibiotic delivery may prevent manifestation of drug resistant in the near future.
Despite that teicoplanin-saturated fibrin discs displayed potential therapeutic activity against the MRSA stain, lyses and absorption of the fibrin sealant [14] were not tested in this study. The absorption of fibrin sealant is relatively important for the antibacterial effect to be prolonged, and we believe that the faster the fibrin sealant absorbs, the more rapid drug will be depleted.
In the previous study, Selçuk Kara mentioned that specific antibiotic only lasts for 48 hours. In our in vitro study show that teicoplanin had continued for 28 days. Of cause, the concentration that we had tested are different despised that they did not mention about the size or thickness of the fibrin sealant. Our study has shown support that the antimicrobial actually could last for 28 days. However, if this in vitro applies in animal or clinical, there are few conditions cannot be ignored. Every single person has differences in DNA, blood supply, etc. Therefore, the expectation antibacterial effect might be shorter as the absorption or lyses effect kick in.
There are limitations in this study. The microorganisms and antibiotic tested in this in vitro mode are not universal. In a further study, more antibiotics and variations in antimicrobial resistance bacteria need to be assessed. In addition, the absorption of fibrin sealant should be tested with the release of antibiotic effect in an animal study. Of course, clinical study should be major consider in the future for more information and data can be a valuable asset in chronic wound healing.
The results of this study show the possibility that antibiotic could be mixed with fibrin sealant to treat wound infections with inadequate of blood supply. Limited concentrations of antibiotics would be mixed with fibrin sealant without over using high-concentration antibiotics. Fibrin sealant may be applied in an infected site, not only for tissue regeneration but also to control an infection [2].
Fibrin sealants provided a prolonged drug delivery, which indicates that antibiotic-saturated fibrin clots act as vehicles to deliver the drug in a local wound infection by controlling the concentration of drug used.